Thermodynamic characterization of the (H2 + C3H8) system significant for the hydrogen economy: Experimental (p, ρ, T) determination and equation-of-state modelling
Second Paper published within the MefHySto Project.
Highlights
- H2-enriched natural gas is a key vehicle to introduce hydrogen in our energy infrastructure.
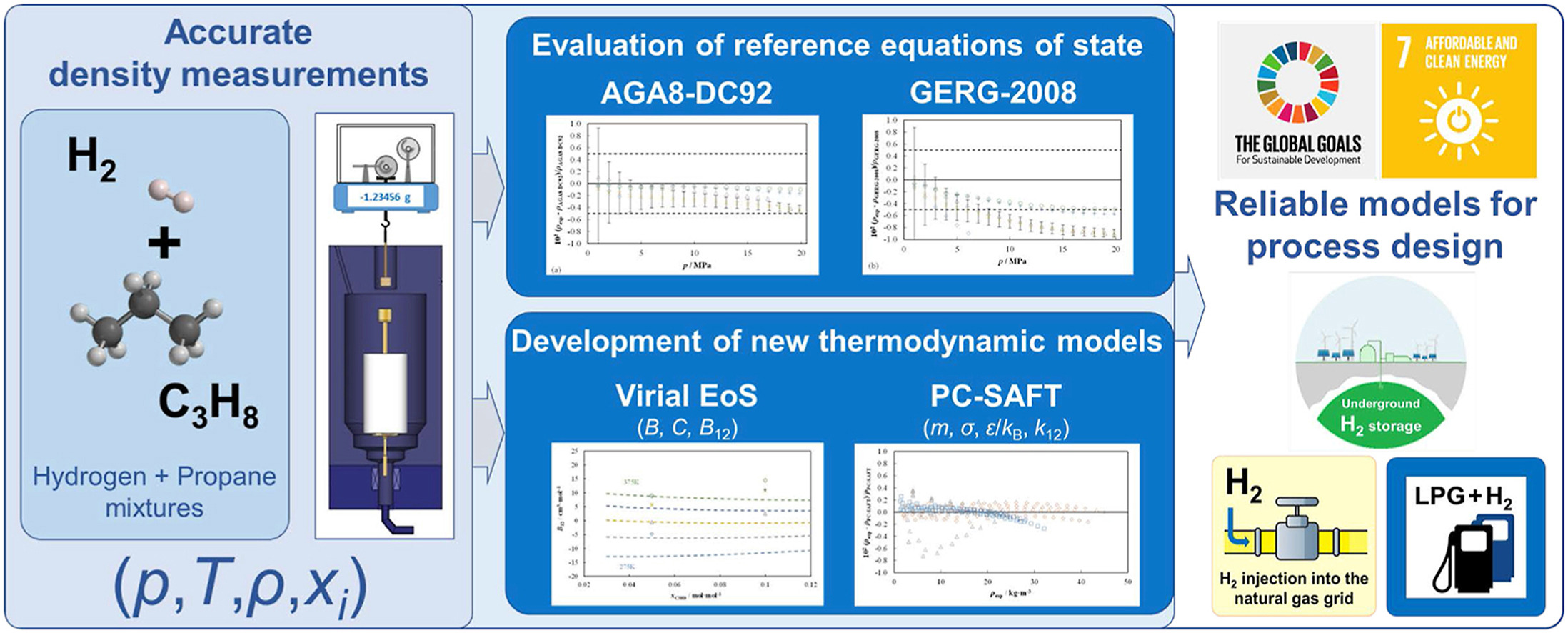
- Three hydrogen-propane mixtures, of reference-quality, were prepared.
- Density of these mixtures was measured over an extended p, T range using a single-sinker densimeter.
- Experimental densities were compared with GERG-2008 and AGA8-DC92 reference equations of state.
- Experimental data were also compared to adjusted virial and PC-SAFT equations of state.
Abstract
For the gradual introduction of hydrogen in the energy market, the study of the properties of mixtures of hydrogen with typical components of natural gas (NG) and liquefied petroleum gas (LPG) is of great importance. This work aims to provide accurate experimental (p, ρ, T) data for three hydrogen-propane mixtures with nominal compositions (amount of substance, mol/mol) of (0.95H2 + 0.05C3H8), (0.90H2 + 0.10C3H8), and (0.83H2 + 0.17C3H8), at temperatures of 250, 275, 300, 325, 350, and 375 K, and pressures up to 20 MPa. A single-sinker densimeter was used to determine the density of the mixtures. Experimental density data were compared to the densities calculated from two reference equations of state: the GERG-2008 and the AGA8-DC92. Relative deviations from the GERG-2008 EoS are systematically larger than those from the AGA8-DC92. They are within the ±0.5% band for the mixture with 5% of propane, but deviations are higher than 0.5% for the mixtures with 10% and 17% of propane, especially at low temperatures and high pressures. Finally, the sets of new experimental data have been processed by the application of two different statistical equations of state: the virial equation of state, through the second and third virial coefficients, B(T, x) and C(T, x), and the PC-SAFT equation of state.


